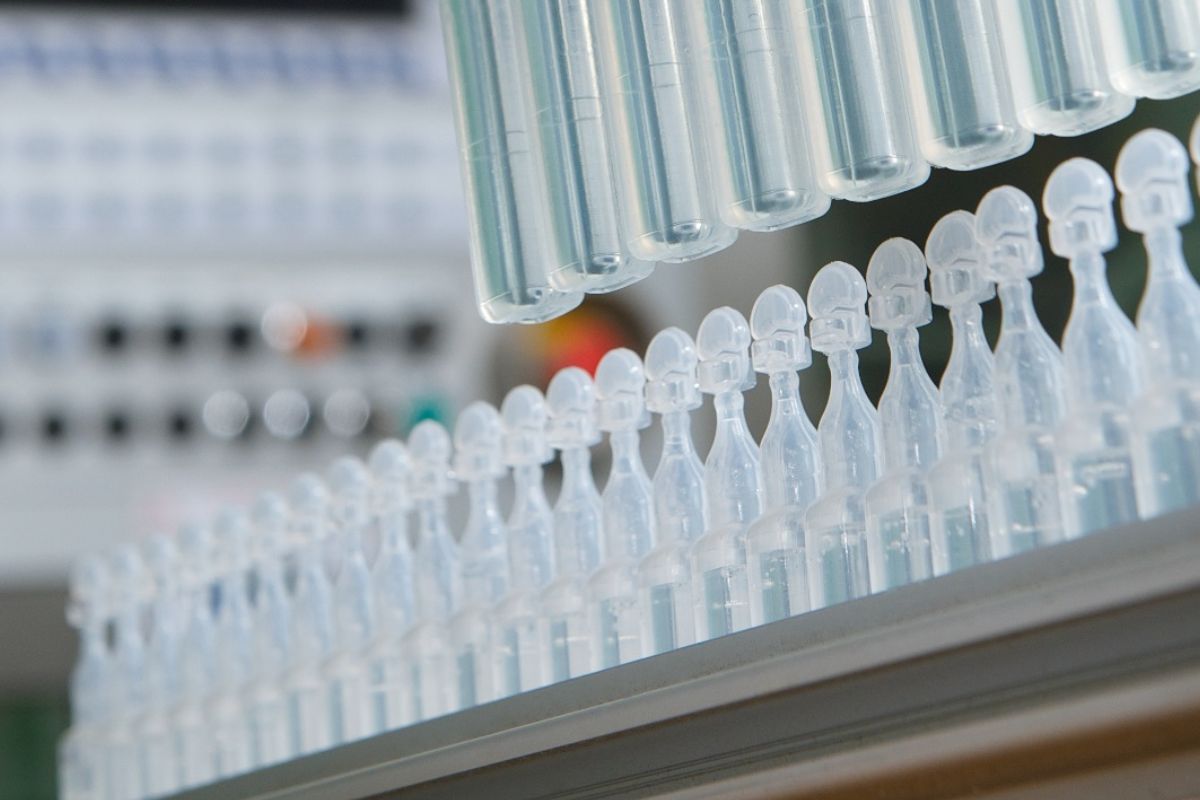
Inhalation therapies require solutions compatible with nebulization, sterile and preservative-free, in order to avoid respiratory irritation and microbiological contamination.
Multi-dose formulations may present hygiene constraints and compatibility limitations with certain active substances.
In this context, ready-to-use single-dose formats are preferred to secure administration and simplify use both at home and in medical environments.