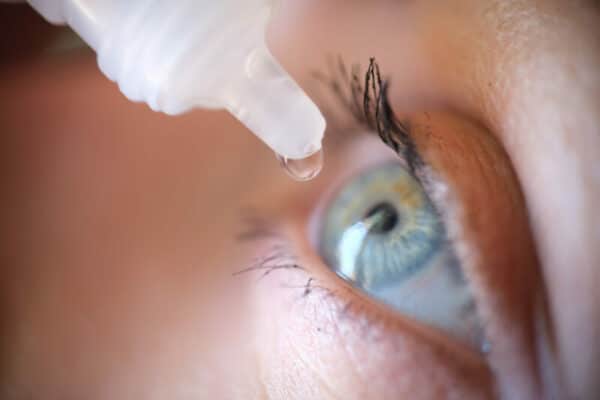
Dry eye and ocular surface irritation require solutions compatible with repeated use and well tolerated.
Preserved formulations may limit instillation frequency and acceptability in certain patients.
In this context, preservative-free medical devices provide an approach suitable for frequent and long-term use.