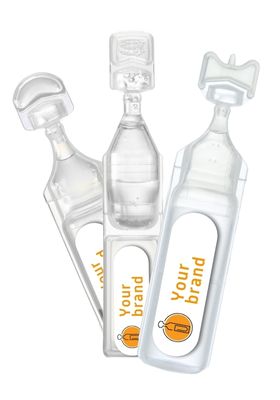
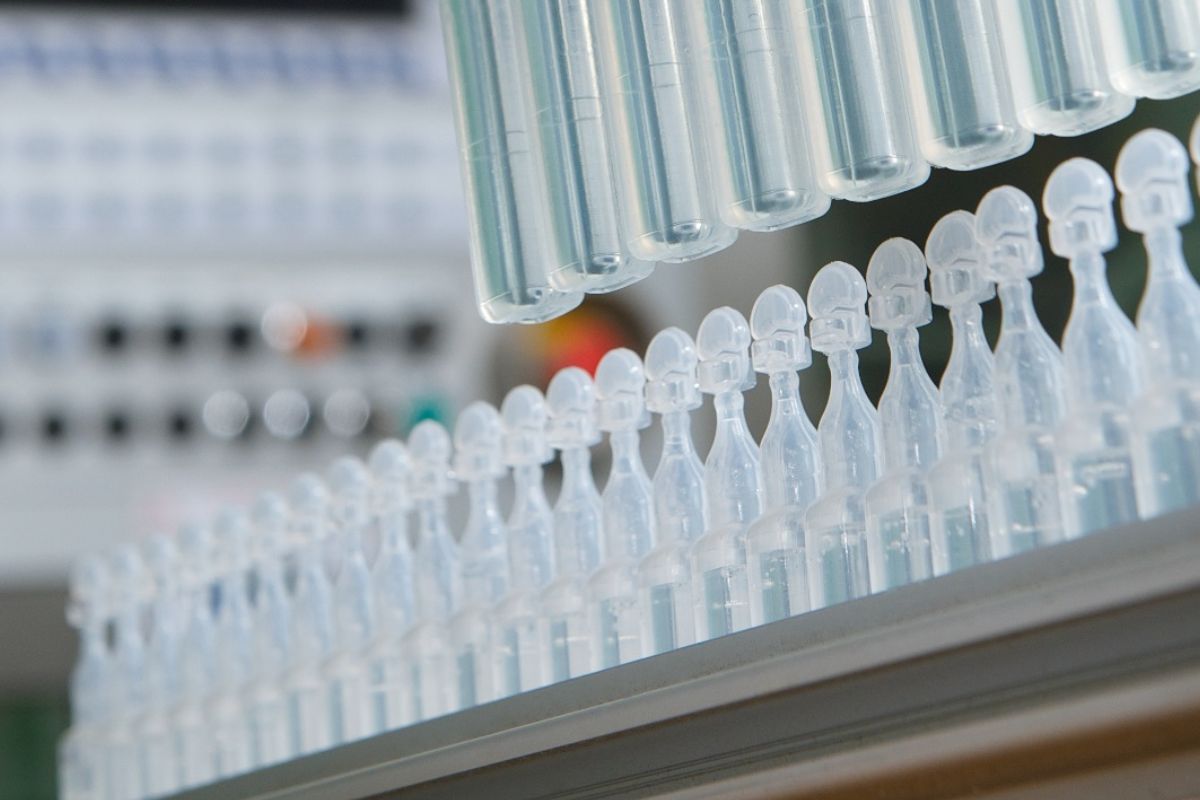
Choosing BFS means choosing simplicity, safety, and performance
- Sterile and preservative-free
- Precise dosage and patient safety
- Portability and ease of use
- Extended shelf life and product stability
- Compatibility with biological and heat-sensitive products